Process flow is a business decision. PAAB has adjusted its requirements for Medical/Regulatory sign-off to respect this fact.
TOPICS
What's changing?
PAAB will no longer require med/reg review to occur prior to initial PAAB submission. Upon submitting, if you have opted not to complete Medical/Regulatory review prior to PAAB review, please click "Not Yet" on the submission form. If Medical/Regulatory review was completed, you will continue to have the option to confirm this and provide the Manufacturer Medical/Regulatory contact.
Why are we making this change?
- We've heard that sequential reviews contribute to delays for some companies when developing APS. A concurrent review by MLR and PAAB may help mitigate this issue.
If a piece is submitted to PAAB and Med/Reg concurrently and Med/Reg requires changes, what will happen to my PAAB submission?
- The decision to run the reviews concurrently is a business decision. If a piece requires significant changes late in the review process or post approval, it will incur a new fee per the current system. This should be considered when making the decision to submit with sign-off or without. As always, we will continue to work closely with the client to minimize delays and help guide them through to the finish line.
What if the manufacturer’s processes require Medical/Regulatory sign-off prior to PAAB submission?
- The new form opens up the client’s options to submit in a manner that suits their comfort level. The form field for the manufacturer’s medical/regulatory sign-off will remain. There is now a new field to select “Not Yet” if there has been no sign-off at the time of submission.
- This is intended to allow flexibility across the industry and organization. There may be pieces the company deems to be “low risk”, such as formulary coverage, where the time to review can be reduced by concurrent review.
What does the new form look like?
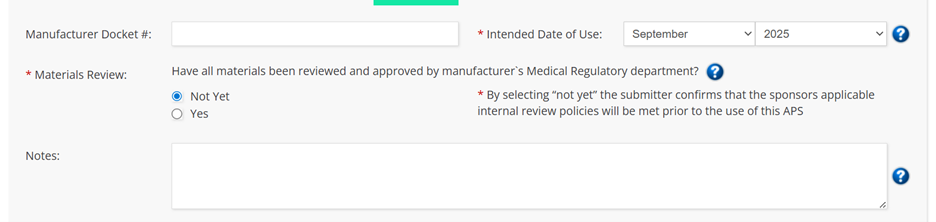
- When you’re submitting a new file, you’ll see the mandatory field of “Material Review” just as you did in the old form. Now, you’ll have two options. If you select “Not Yet”, copy stating “By selecting “not yet” the submitter confirms that the sponsor’s applicable internal review policies will be met prior to the use of this APS.” will pop up. This is the understanding PAAB expects of the submitter.
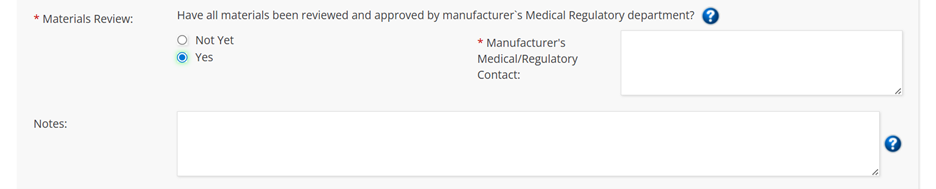
- If the submitter selects “Yes”, a box to fill in the manufacturer’s Medical/Regulatory contact appears as per the old form (see below).
Does Medical/Regulatory need to sign-off at all?
- Yes – Per Code section 1.6B (updated April 2025)
- 3. The submitter must provide confirmation within the submission form indicating that a sponsor Company Official from the Medical, Regulatory, or Compliance department will grant approval prior to use.
It’s important that an official from medical or regulatory from the sponsor’s organization is still within the loop for advertising put out into the market. This change simply allows for the business decision of “when” during the content creation process they are involved. It allows for flexibility while maintaining accountability.

